Delta2000 is one of the largest national and international manufacturers of clean rooms or cleanrooms.
What are cleanrooms and what are they for? What are their main guidelines?
In this article we will analyse all these aspects in detail, answering some questions that often arise when talking about cleanrooms: what are cleanrooms? What are their main guidelines? Why invest in a cleanroom today?

Clean rooms: a brief definition and some interesting facts about their origin
What is a cleanroom?
A cleanroom, from the English ‘cleanroom’, is a contamination-controlled environment. The purpose of the cleanroom is to provide a working environment that limits the presence of particles/particulates within it thanks to a special air filtration system.
Where do clean rooms originate and what are their origins?
The cleanroom originated in the hospital environment and was only later exploited in the industrial environment by those companies that needed to keep contamination levels of their products under control.
It should be remembered that the presence of micro-organisms in the environment can alter the properties of certain products, so working in non-sterilised environments – especially in the case of foodstuffs (meat, fish, dairy products, vegetables or pre-cooked products) – can put people’s health at risk, causing poisoning.
All cleanrooms produced by Delta2000 are designed for use in contamination-controlled areas and are intended for multiple sectors: Pharmaceutical, Biomedical, Chemical, Cosmetic and Food.
Not forgetting structured projects for the hospital sector. Sector, as anticipated, of origin of the same.

Cleanrooms and structural requirements
During the production process of a cleanroom, it is of paramount importance to comply with certain structural requirements, including the following:
- materials must not release particles;
- surfaces must be smooth and easy to clean;
- fittings must have rounded edges;
- sockets and fixtures must be coplanar;
- pipes must run outside the premises.
Particular attention must also be paid to the air treatment system. This prevents microorganisms or dust from entering from the outside and thus keeps the interior environment completely sterile and aseptic.

One of the main sources of contamination within the cleanroom is the production process together with the machinery. Care must be taken with the handling of products, cleaning and maintenance of the cleanroom itself.
But not only that, even humans, through their own movement such as walking, can emit millions of particles that pose a danger to a cleanroom (particle contamination).
In fact, the personnel entering the cleanroom, in addition to being carefully trained, must wear sterilised or disposable clothing, following a precise sequence.
Furthermore, this dressing ritual must take place in a changing room close to the clean room that avoids contamination to the outside.
Delta 2000 produces a wide variety of lockers and benches for cleanroom dressing areas to avoid cross-contamination.
DeltaFURNITURES lockers and benches are designed to position garments, such as protective gowns and sterile footwear, used within aseptic environments. By placing gowns and footwear inside them, problematic contamination can be avoided within sterile areas.
By meeting the precise purity standards required, it is possible to guarantee completely sterile production.
Cleanroom guidelines: the UNI EN ISO 14644 standard
The reference guidelines for clean rooms are: the UNI EN 14644 standard, which in Europe has replaced the American Federal Stardards, and Good Manufacturing Practice (GMP).
The essence of the standard is the table below. The table shows the maximum concentrations of particles contained in one cubic metre of air, in relation to the different cleanliness classes.
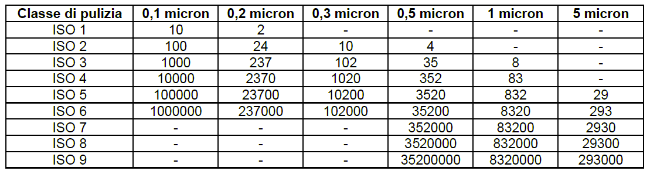
In order to know whether a cleanroom is optimal for your facility you need to understand, based on the sector you operate in, what standard it must meet. Cleanrooms are classified according to the level of cleanliness of the air inside them, and based on the amount and size of particles per volume of air there is the class to which the room conforms.
The old Federal Standard 209 was withdrawn in 2001 and officially replaced by ISO-14644-1 in 1999, but is still widely used.
This standard has classes ISO 1 to ISO 9, starting with ISO 1 which is the ‘cleanest’ class up to ISO 9 which is the ‘dirtiest’ class. After ISO 9 there are the Non-Classified (N.C) environments.
Why invest in a Cleanroom

Not only is the use of cleanrooms gaining in importance in today’s world, but we are also seeing an expansion into new areas of application. Whereas originally only the microelectronics and semiconductor sectors embraced this type of idea, today even the food production, optical, pharmaceutical-medical and aerospace industries through to injection moulding companies and research institutes are choosing to invest in a cleanroom.
As the economy develops, increasingly high-tech and high-precision industrial processes are in demand. The use of cleanrooms is the perfect answer to these new requirements, not only in relation to product quality, efficiency and smoothness of the production process, but also in relation to the very high health and comfort of operators.
Rely on Delta2000 when choosing your All Inn solution for clean rooms and sterile environments.
Certified solutions, quality and adherence to project deadlines, to meet every customer need.
Request information
Are you interested in receiving information about our products?
Fill out the form and our Sales Team will be in touch as soon as possible.




