Designing a pharmaceutical cleanroom is one of the most complex and regulated engineering processes ever.
Every choice, from wall panel type to filter classification, from airflow management to material selection, has a direct impact on finished product quality, regulatory compliance, and operator safety.
This guide was produced by Delta 2000, a company based in Cavenago Brianza (MB) and a leading international partner in the design, manufacture and installation of modular cleanrooms. In more than two decades of experience in the pharmaceutical, medical, electronic, food and cosmetic industries, we have acquired in-depth knowledge of every aspect related to controlled contamination environments.
You will find in this guide everything you need to know to properly approach a cleanroom project: from ISO 14644 to European GMP guidelines (EU GMP Annex 1), from the choice of structural components to qualification checks.
An article designed for technical managers, process engineers, quality managers and facility managers who want to explore the topic with rigor and clarity.
1. What is a cleanroom and why it is essential in the pharmaceutical industry
A clean room (in English clean room) is a confined and controlled environment in which the concentration of airborne particles, dust, microorganisms, and chemical aerosols is kept within precise limits through air filtration systems, differential pressurization, inert materials, and strict operating protocols.
In the pharmaceutical industry, a clean room is not an option-it is a regulatory requirement. The production of sterile drugs, medical devices, active ingredients (APIs) and injectable products requires environments in which microbiological and particle contamination is reduced to near-zero values.
Even the slightest contamination can compromise the effectiveness of the drug, cause adverse reactions in patients or result in the recall of entire production batches.
The consequences of a poorly designed or noncompliant cleanroom are serious: penalties from regulatory authorities (AIFA in Italy, EMA in Europe, FDA in the US), suspension of manufacturing approvals, public health risks, and irreversible reputational damage to the company.
The main pharmaceutical applications of clean rooms:
- Production of sterile drugs (vialoids, lyophilized, injectable solutions)
- Aseptic filling and processing of active ingredients
- Primary packaging of sterile products
- Advanced therapy drug manufacturing (ATMP)
- Microbiological quality control laboratories
- Production of implantable medical devices (class II and III)
2. The reference standard: ISO 14644 and EU GMP
Anyone who wants to design, build, or operate a cleanroom must contend with a complex international regulatory framework.
The two main families of standards governing this area are the ISO 14644 series and the European Union Good Manufacturing Practice ( GMP) guidelines.
ISO 14644: the international cleanroom standard
The ISO 14644 series is the international reference standard for the classification, design, monitoring and management of cleanrooms and contamination-controlled environments. It consists of several parts, each of which deals with a specific aspect:
- ISO 14644-1: Classification of air cleanliness (ISO classes 1 to ISO 9).
- ISO 14644-2: Monitoring to demonstrate continued compliance with ISO 14644-1
- ISO 14644-3: Test methods for clean rooms.
- ISO 14644-4: Design, construction and commissioning.
- ISO 14644-5: Operational management of clean rooms.
- ISO 14644-10: Classification of surface cleaning by chemical concentration.
EU GMP Annex 1: the European standard for sterile medicines
For the European pharmaceutical industry, the most relevant regulatory document is GMP Annex 1 (Manufacture of Sterile Medicinal Products), which in its updated version of 2022 introduced significant changes in the way sterile manufacturing environments are classified and managed.
GMP Annex 1 classifies production areas into four grades (A, B, C, D), each corresponding to specific limits of particulate and microbiological contamination, both at rest (at rest) and operational (in production) conditions.
One of the most innovative aspects of Revision 2022 is the introduction of the concept of Contamination Control Strategy (CCS): a systematic and documented approach to controlling all potential sources of contamination, which complements, but does not replace, ISO classification.


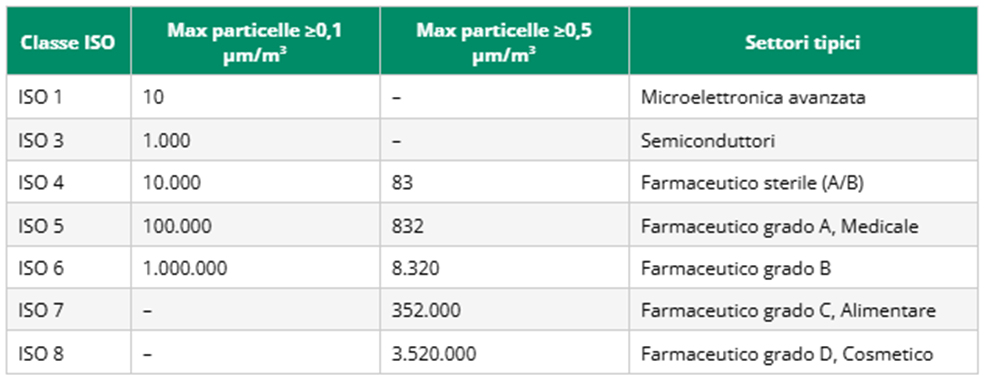
3. The classes of cleanliness: from ISO 1 to ISO 9
The ISO classification defines the level of particulate contamination permissible in a confined environment.
Classes range from ISO 1 (the cleanest, almost particle-free) to ISO 9 (the least restrictive, comparable to ambient air under normal conditions). In the pharmaceutical industry, ISO classes 4, 5, 6, 7 and 8 are mainly worked with.
The following table summarizes the main ISO classes with their particle concentration limits and typical industrial applications:
Click on the table to enlarge it
Note: the limits given refer to the maximum number of particles per cubic meter of air. Grades should be checked periodically according to ISO 14644-2 under both at rest and operational conditions. For GMP grades A/B/C/D, refer to the specific table in EU GMP Annex 1.
4. The structural components of a cleanroom
A clean room is not simply a room with white walls. It is an integrated system of architectural, plant engineering and technological components that must work in coordination to ensure the required cleanliness class. Let’s look in detail at the main structural elements.
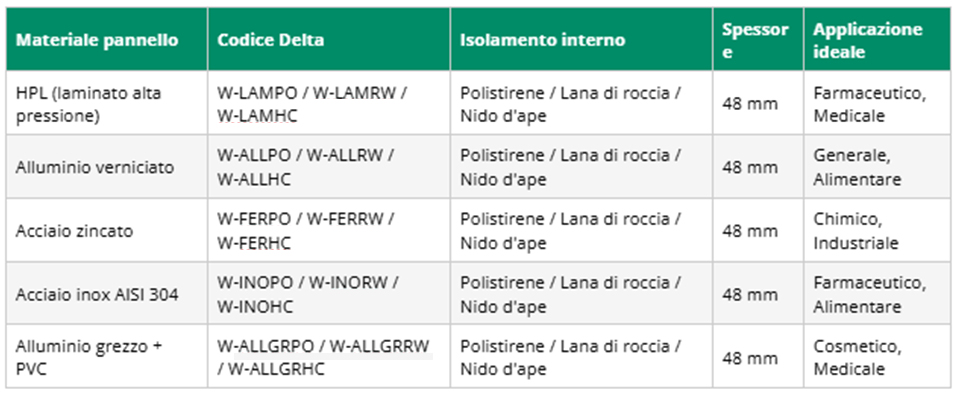
4.1 Modular Walls: DeltaWALL™
Walls represent the physical perimeter of the controlled zone and must meet precise requirements in terms of airtightness, ease of cleaning, chemical resistance and fire behavior. Delta 2000 produces five families of DeltaWALL™ wall panels, each designed for specific applications:
Click on the table to enlarge it
All DeltaWALL™ panels have standard thickness of 48 mm, maximum dimensions of 1286 mm × 3030 mm (for HPL) and minimum dimensions of 90 mm × 90 mm. The jointing system is by extruded aluminum profiles with 4 mm silicone to ensure airtightness. Panels are available in standard colors (Sand, Silver Blue, Spring White) or upon request in the entire RAL range.
TECH TIP: AISI 304 (scotch-brite) stainless steel panels are the preferred choice for grade A/B pharmaceutical environments where frequent sanitization with aggressive chemicals is required. The scotch-brite finish provides ease of cleaning and corrosion resistance.
4.2 Ceilings: DeltaCEILING™
The suspended ceiling is the component through which filtered air from the HVAC system is distributed. Delta 2000 manufactures two types of suspended ceilings DeltaCEILING™: the T-grid system with Omega 65 profiles and the walkable ceiling.
The T-grid system is the most widely used in pharmaceutical cleanrooms: it involves the housing of HPL panels (5 mm thick), sandwich aluminum (6 mm) or aluminum with polystyrene (20 mm), interspersed with HEPA/ULPA filters and LED lighting modules. The walkable ceiling, on the other hand, is suitable for overhead installations that require maintenance access to the plenum.
Both systems are designed to ensure uniform air flow, reduce dead spots, and facilitate cleaning and sanitizing.
4.3 Doors: DeltaDOORS™
Cleanroom doors must combine the physical barrier function, to maintain differential pressurization, with practicality of use in high-frequency transit operating environments.
The DeltaDOORS™ provides doors that can be installed on both DeltaWALL™ panels and existing walls in the configurations:
- Single door (1 leaf) – Ref. D-W1.0 / D-MW1.0
- One and a half door (1.5 doors) – Ref. D-W1.5 / D-MW1.5
- Double door (2 doors) – Ref. D-W2.0 / D-MW2.0
- Door with frame on 4 sides – Ref. D-W4S / D-MW4S
- Single Sliding Door – Ref. D-SL1 / D-MSL1
- Double Sliding Door – Ref. D-SL2 / D-MSL2
Sliding doors are the preferred solution for high-grade cleanrooms: they eliminate air movement generated by opening swinging doors, reducing the risk of turbulence and particle ingress from outside.
4.4 Windows and visuals: DeltaWINDOWS™
Windows and viewports are key elements for visual supervision of production environments without direct access.
The DeltaWINDOWS™ includes 48 mm thick windows mountable on DeltaWALL™ panels, with double-chamber thermopan glass (12 mm thermopan glass), telescope windows and variants with panic glass (safe crash). The flush-wall frame ensures no corners and easy cleaning.
4.5 Flooring: DeltaFLOOR™
The floor of a cleanroom should be seamless, resistant to chemicals used for sanitation, antistatic (ESD) or conductive where required, and easily inspected.
Delta 2000 offers solutions in epoxy resin or PVC homogeneous with integrated skirting (DeltaPROFILE™ coving), eliminating corner joints that are areas of dirt and microorganism accumulation.


5. HVAC systems and air filtration
The air handling system is the technical heart of any clean room. HVAC (Heating, Ventilation and Air Conditioning) includes all the systems that control temperature, relative humidity, differential pressure and, most importantly, the concentration of particles in the ambient air.
HEPA and ULPA Filters
Air quality in pharmaceutical clean rooms is ensured by high-efficiency filters installed in the ceiling or end diffusers. The two main standards are:
- HEPA (High Efficiency Particulate Air): efficiency ≥99.97% for 0.3 µm particles. Used for ISO classes 5-8 and GMP grades C-D.
- ULPA (Ultra Low Penetration Air): efficiency ≥99.9995% for 0.12 µm particles. Used for ISO classes 3-4 and GMP grades A-B.
Air replacements and pressurization
The number of air changes per hour (ACH) depends on the ISO class required and the activity in the environment. For an ISO class 7 (GMP grade C), 20-40 ACH are typically required; for an ISO 5 (GMP grade A), values in excess of 240 ACH are reached with unidirectional flow.
Positive differential pressurization (the clean room is at higher pressure than adjacent rooms) prevents the entry of unfiltered air. The pressurization cascade must be carefully designed, especially in areas with multiple adjacent GMP grades.
GMP NORMATIVE PARAMETER ANNEX 1: For grade A GMP environments, the standard requires unidirectional air flow (laminar flow) with velocity between 0.36 and 0.54 m/s measured in the work plane. This condition ensures that particles are continuously and uniformly moved away from the critical area.
6. Materials and surfaces: the requirements of the regulations
The choice of materials is one of the most critical aspects in the design of a pharmaceutical cleanroom.
Every surface (walls, floor, ceiling, doors, furniture) must meet precise regulatory and technical requirements.
The basic selection criteria are:
- Absence of particles: materials must not release dust, fibers or particles into the environment. Porous materials, textiles or coatings that flake off are excluded.
- Chemical resistance: surfaces must resist disinfectants and sanitizing agents used in routine cleaning (sodium hypochlorite, acetic peracid, isopropyl alcohol and others).
- Ease of cleaning: no sharp edges, grooves or open joints that could retain dirt or microorganisms. Curved (coving) corner fittings are prescribed by GMP standards.
- Fire behavior: materials must meet at least fire class B-s1-d0 for exterior cladding (such as HPL laminate of DeltaWALL™ panels).
- Mechanical strength: surfaces must resist impact and wear from the use of carts, pallet trucks and production equipment.
I DeltaWALL™ HPL panels meet all these requirements: impact resistance above IP > 150 (EN 438-2.10), scratch resistance grade > 3 (EN 438-2.25), formaldehyde release class E1 and fire reaction class B,s1-d0.
7. Flow design: personnel, materials and air
One of the most overlooked, and most critical, aspects of cleanroom design is flow management: who enters, how they enter, what they bring in, and how the air moves through the environment. Poorly thought-out flow design can negate the investment in high-quality components.
Personnel flow
Personnel are the main source of biological contamination in a cleanroom. Each worker continuously emits particles (skin cells, hair, respiratory droplets) that must be intercepted before entering the controlled area.
The dressing course typically involves:
- Shoe changing area (disinfection mat)
- Changing room with DeltaFURNITURES™ lockers for personal clothing
- Dressing area for sterile clothing (coveralls, shoe covers, gloves, masks)
- Air shower or decontamination chamber before entry
- The lockers and the benches DeltaFURNITURES™ are made of fire retardant melamine or full HPL, with customizable dimensions, and are the ideal solution for locker rooms adjacent to clean rooms.
DeltaPASSBOX™ material flow and passboxes.
Transferring materials between differently classified environments is one of the most delicate operations.
The use of a passbox (pass-through) allows transfer without operators having to cross physical barriers between one GMP grade and another.
The DeltaPASSBOX™ system is available in static (without an active decontamination system) and dynamic (with electromechanical door interlock, which prevents both doors from opening at the same time) versions. It can be installed on the wall (bridge pass-through) or on the floor, with customizable internal dimensions.


8. Delta 2000 modular solutions for the pharmaceutical industry.
Delta 2000 specializes in turnkey supply of modular cleanrooms for the pharmaceutical industry, from needs analysis to design, production to installation and after-sales service.
The DeltaWALL™ modular system offers concrete advantages over traditional construction:
- Quick installation: modular panels are assembled on site without masonry work, drastically reducing production downtime.
- Flexibility and scalability: the modular system allows the cleanroom to be reconfigured or expanded without demolition, simply by disassembling and reassembling the panels.
- Certified quality: all components are manufactured in the factory under strict quality control, ensuring uniformity and repeatable performance.
- Regulatory compliance: the system is designed to meet ISO 14644 and GMP Annex 1 requirements, with full technical documentation to support validation.
- Specialized technical support: the Delta 2000 team supports the client at every stage of the project, from defining functional requirements to plant qualification (IQ/OQ/PQ).
Markets served include Italy, Europe, the United States, Canada, and South America, with a network of international partners able to provide local support.
9. Validation and qualification: IQ, OQ, PQ
A pharmaceutical cleanroom is not operational until it has been formally validated and qualified. The qualification process has three sequential steps, according to GMP terminology:
- IQ (Installation Qualification): verifies that all components have been installed correctly, according to supplier specifications and design requirements. Includes inspection of all structural, plant and control components.
- OQ (Operational Qualification): verifies that the cleanroom is functioning properly in all its operating modes. Includes measurement of particle concentration under at rest conditions, verification of HVAC parameters (air flow rate, hourly changes, differential pressures, temperature and humidity), HEPA/ULPA filter leakage tests (DOP test or PAO test).
- PQ (Performance Qualification): verification that the cleanroom maintains the required performance during normal production operations (operational conditions). Includes environmental microbiological monitoring, verification of particle concentration during production, and assessment of the impact of operator activities.
Delta 2000 supports its clients at all stages of qualification, providing the necessary technical documentation (URS, DQ, project specifications) and collaborating with Quality Assurance teams in drafting IQ/OQ/PQ protocols.
10. Common mistakes to avoid in the design phase
Our experience from hundreds of installations has enabled us to identify the most common mistakes companies make when designing a pharmaceutical cleanroom.
Avoiding them at the preventive stage can mean the difference between a successful project and costly corrective action.
- Undersizing the ISO class: choosing a lower class than actually needed, in order to keep initial costs down, almost always leads to noncompliance at qualification and having to redo significant parts of the plant.
- Ignoring staff flow: designing the cleanroom without defining the staff dressing and decontamination paths is one of the most serious mistakes and difficult to correct after the fact.
- Underestimate pressurization: a poorly designed pressurization cascade can create cross-contamination risk zones, especially between areas of different GMP grade.
- Choose unsuitable materials: the use of materials that are not certified or resistant to sanitizing agents compromises the integrity of surfaces over time, creating crevices and areas of microbial accumulation.
- Neglecting preventive maintenance: a cleanroom requires a rigorous preventive maintenance program-periodic filter replacement, recalibration of measuring instruments, seal check-without which performance rapidly degrades.
- Don’t involve the supplier from the project phase: relying on Delta 2000 as early as the functional requirements definition and preliminary design phase allows you to optimize technical solutions and anticipate critical issues.
11. Conclusions: designing well to produce better
The design of a pharmaceutical cleanroom is a process that requires multidisciplinary skills, in-depth knowledge of international regulations, and a systems approach that considers the manufacturing environment as an integrated set of structural, plant, and procedural components.
At Delta 2000, we have built over time a design method that always starts with an analysis of the customer’s requirements, the ISO class needed, the GMP grade required, the area of application, and the operational needs, to arrive at a tailored solution that combines regulatory compliance, construction quality, and economic efficiency.
The DeltaWALL™ modular system, together with the DeltaCEILING™, DeltaDOORS™, DeltaWINDOWS™, DeltaFLOOR™, DeltaFURNITURES™, and DeltaPASSBOX™ solutions, is one of the most comprehensive and versatile offerings on the international market today for cleanroom implementation in every industry and for every ISO classification level.
Do you have a cleanroom project to implement?
Our technical consultants are available to analyze your needs, define the correct ISO classification and design the modular solution that best suits your industry.
Contact a Delta 2000 consultant → www.delta-2000.com | info@delta-2000.com | +39 02 9501688