Delta2000, a global player in cleanroom manufacturing, answers some of the most frequently asked questions about controlled contamination environments.
Read our selected FAQs on the topic of clean rooms.
White Rooms FAQ:
- What is a clean room and in what areas is it needed?
- How does a clean room work?
- How do pressure cascades prevent contamination in the cleanroom?
- What are airflows and why are they important in cleanrooms?
- What standards apply to cleanroom classification?
- What are the cleanroom classes according to EN ISO 14644?
- What are the cleanroom classes according to the EU GMP?
- What should be considered when assembling a cleanroom?
- What transport equipment is suitable for clean rooms?
- Request information
Some industries (such as pharma, biomed, chemical, cosmetic, food, and hospital-health) are held to stringent hygiene requirements that go far beyond routine cleaning.
Since these are sensitive processing environments, the concentration of airborne particles dispersed in the room air must remain within a well-defined limit: an environment that meets this characteristic is called a clean room or cleanroom.
Depending on the industry, there are international standards that strictly regulate cleanrooms. Thus, there are differences between a food cleanroom, a pharmaceutical cleanroom or a hospital cleanroom.
The standards have rules on several aspects of clean rooms:
- The concentration of particulate matter per cubic meter;
- The ability to filter and clean the air;
- The type of equipment, materials, and work machines that can be accessed;
- The appropriate means of transportation to enter cleanrooms;
- The clothing of staff entering and leaving clean rooms.
Also read this article to learn more about this topic: The importance of clean rooms today, in manufacturing environments where cleanliness must be total
1. What is a clean room and in what areas is it needed?

Clean rooms produced by Delta2000 are workplaces where the concentration of airborne particles is kept as low as possible, by artificial means. In this way, highly sensitive production techniques, or laboratory research and testing, can take place under the hygienic conditions required by law. If necessary, sterile working environments can be created.
Aseptic chambers are necessary in some industries because the air always contains particles composed of dust, gases, pollen, and other particulates. In manufacturing environments where food, drugs, cosmetics but also, for example, computer chips are handled, these particles can be harmful.
In contrast, inside the cleanroom, the concentration of microparticles is extremely low, so that the risk of contamination or damage is drastically lowered and the necessary technical and hygienic conditions are thus met.
You must always plan to set up a cleanroom for:
- Environments devoted to food production and processing;
- Environments devoted to semiconductor manufacturing and information technology;
- Aerospace technology sector;
- Performance of laboratory tests:
- Environments where medical, biological and chemical research projects are conducted;
- Optics and laser technology sector;
- Environments devoted to the production of pharmaceutical products.
2. How does a clean room work?
A clean room is not always a room or a chamber. With appropriate isolation measures, you can also set up entire sections of a building as clean rooms. For example, this is the case with the aseptic cleanroom designed by Delta2000, which is capable of forming a self-contained unit in which the concentration of particles can be precisely measured and monitored. In addition, you can control other parameters such as humidity, air pressure, and temperature. The conditions in an aseptic chamber must be constant.
The cleanroom assembly phase therefore is crucial. Delta2000 installs very powerful ventilation and air conditioning systems that ensure air exchange according to the required standards.
A technology called positive-pressure ventilation which generates a flow of air that pushes particulate matter out of the room. When there are special safety requirements to be met (e.g., if harmful chemicals or pathogens are being worked on in the cleanroom), then negative pressure ventilation, which sucks up particles in the air, purifying it.
3. How do pressure cascades prevent contamination in the cleanroom?
In many areas, cleanroom air is kept “clean” by pressure cascades. The air inside the cleanroom thus maintains a low content of suspended dust microparticles.
Cleanrooms are classified by taking into account the pressure exerted and the amount of particles per cubic meter that are airborne. Physically, the higher the pressure prevailing in the room, the higher the purity of the cleanroom air. The ISO1 cleanroom standard has the highest pressure: that is, it means it achieves the highest possible level of air purity.
To achieve additional protection from potentially harmful agents or contaminants, additional modes of air filtration can be used.

Thegoal of ventilation technology is to eliminate airborne particles generated during work performed inside the cleanroom. However, employees, transport equipment and work materials, which come from outside and enter inside the cleanroom, are also a high contamination risk factor.
To remedy the problem, the airlock is used for personnel and all materials entering the cleanroom. Airlocks remove the residual harmful agents left on employees, equipment, and materials before entering the cleanroom. The action is carried out by a strong air flow.
Instead, shoe soles, trolley wheels and other objects in contact with the ground are decontaminated through special mats with antibacterial adhesive. In addition, almost always to enter a cleanroom, staff must wear a specific suit or sterile clothing.
Delta2000 devotes much attention to the study of locker rooms and/or dressing/dressing areas by designing lockers e benches, so as to achieve a functional environment that is best integrated with the overall cleanroom structure.
4. What are airflows and why are they important in cleanrooms?
People, objects, and work processes inside the cleanroom can emit contaminating microparticles. Ventilation technology eliminates these airborne substances.
The level of purification depends on the industry and the standard of the cleanroom: on this basis, a ventilation technology with specific requirements is chosen. Ventilation may simply reduce the concentration of particles through induction of clean air, or more complex processes such as extracting air masses from the interior and replacing them with clean air (as free of suspended particles as possible) may be required.
Within the cleanroom, air flows to prevent contamination can be:
- turbulent
- laminar (ordered and uniform, suffering no deflections from any obstacle and moving at a uniform speed).
Laminar airflows are preferred when it is necessary to keep the air as particle-free as possible: in fact, compared with turbulent airflows, they manage to achieve almost 10 times more effective air decontamination.
5. What standards apply to the classification of clean rooms?
The industrial sector and the operations that are carried out inside the cleanroom determine very different rules regarding the tolerability of the presence of particles or pathogens. Cleanrooms therefore are classified on the basis of strict cleanliness requirements, depending on use and industry.
For example, in the semiconductor or space technology sector, a clean room protected from chemicals and airborne microparticles that could ruin components is required. Conversely, in the food industry, a food cleanroom is required where the amount of microorganisms present is also kept as low as possible. Bacteria are also the enemies of cleanrooms in the biomed and pharma areas.
The classification of clean rooms is regulated by specific norms from which international standards, valid in all states, have been obtained:
- UNI EN ISO 14644-1: This standard falls under UNI EN ISO 14644. It regulates the production of clean rooms and associated controlled environments based on air cleanliness classification. The cleanliness classes range from ISO 1 to ISO 9. Maximum particulate matter ceilings are established for each class. It also replaces the previous US 209E federal standard.
- UNI EN ISO 14159: “Safety of machinery – Hygiene requirements for machinery design.” The standard regulates hygiene requirements for the design of machinery and transport equipment.
- VDI 2083: This is a technical guide edited by VDI, the German Association of Mechanical Engineers. It details guidelines for the design and installation of ventilation and air conditioning systems in buildings to meet the UNI EN ISO 14644 quality standard.
- Guide to GMP: GMP is Good Manufacturing Practices”. The Guide, in ‘Appendix 1, indicates the manufacturing standards for sterile medicines. These are classified into 4 cleanroom classes from A to D. Adopting these measures ensures particle and contamination control during the production of medicines.

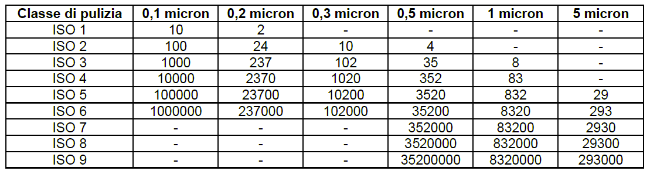
6. What are the cleanroom classes according to EN ISO 14644?
There are 9 classes of clean rooms of the ISO standard. For each class, the following are measured:
- particle size;
- No. plots / mc.
ISO classes 1 to 3 are the so-called clean rooms: these are the most effective, suitable for achieving highly controlled environments where a high degree of purity is required.
- Max particle size: 0.5 µm
- The concentration of particles in these environments is so low that they do not pose a problem of contamination or damage to sensitive products.
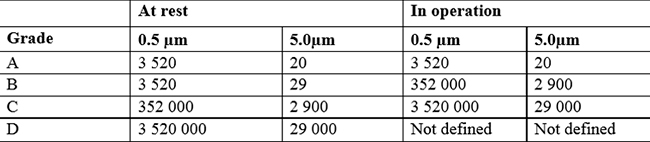
7. What are the cleanroom classes according to the EU GMP?
Instead, the GMP guidelines classify 4 groups of clean rooms, from A to D, where class A represents the highest degree of air cleanliness to meet more stringent requirements. Class D, on the other hand, is the most basic level, applicable in settings with less stringent requirements.
It is mainly the pharmaceutical and medical sectors that use this classification, which then also takes into account microbiological air contamination (bacteria, viruses, microorganisms, pathogens).
GMP classification is based on the concentration of airborne particles, both at rest and during operation of cleanroom cleaning systems.
The limit values given in the GMP guidelines are a recommendation. The mandatory requirements are those set by laws and international standards for each industry. Through these strict regulations, we have the assurance that the products processed within the cleanrooms are safe for consumption and meet the quality and safety requirements needed in such sensitive industries.
Equipment and materials that are brought into a cleanroom from outside must also pass through “material closures” in order to maintain the level of cleanliness indicated by the specific requirements. For cleanroom classes ISO 1 through ISO 3, the requirements for air purity, tools, and materials that can enter are the strictest. In the highest security regimes, certain equipment such as forklifts are prohibited: the only way to get materials into a highly secure cleanroom is by manual transport.
8. What should be considered when assembling a cleanroom?
When assembling a clean room, it is good to limit the furniture and the amount of items to the essentials to avoid contamination. The utensils and furniture should, however, be made only of abrasion-resistant materials to avoid the detachment of potentially harmful particles. This is why, for example, stainless steel cabinets or work tables with melamine coating are used.
In addition, furniture, i floors and wall coverings must be crack-resistant, smooth and seamless to avoid unsanitary deposits of particles and dust. Again, materials that are easy to clean and disinfect must be used.
Attention must be paid to every detail, including work chairs that must be made of antistatic materials and have only closed and smooth surfaces, or they must be properly sealed with a layer of plastic film. Every material that goes into clean rooms must comply with these logics, including the most mundane items such as notebooks, pens, notepads, etc.
Cleanrooms cannot be accessed without meeting cleanliness requirements. Very often you go through areas that are separate from each other, cleanrooms that are increasingly efficient in minimizing contamination.
Conversely, on the way out, if you are working in contact with hazardous substances, a process of disinfection of clothing and materials is carried out. This is another reason to provide dispensers with disinfectant.
Adequate signage should be installed within each cleanroom environment, especially where there are possible sources of danger, to ensure proper workplace health and safety for staff.

9. What transport equipment is suitable for clean rooms?

After the cleanroom spaces have been properly set up, it is also important to choose permissible transport equipment (pallet trucks, carts or transport trolleys).
Transport vehicles move not only inside the clean rooms, but also outside and in multiple operational areas. Therefore, they may have residual harmful particles that increase the risk of contamination. Transport equipment for cleanrooms must comply with the UNI EN ISO 14159 standard.
Among the materials to be preferred is definitely stainless steel, which is resistant to chemicals and abrasion, acids, alkalis, and moisture. This material should be smooth. In the case of stainless steel pallet trucks and forklifts, the fork tips, if kept closed, can protect goods from airborne particles. Stainless steel conveying equipment also has sliding nylon rollers with adequate resistance to abrasion during movement.
There are special transport carts, enclosed and made of stainless steel, used to transport and store decontaminated components from inside cleanrooms. Being enclosed they protect the goods from dust and particulates. For added security they can be equipped with electronic locks.
Not only that, there are transport carts that have their own air filtration system: it sucks in the indoor air and with a pre-filter function and a HEPA filter cleans the outdoor air. These special transport vehicles are also useful for cooling products.
10. Request information
Are you interested in receiving information about one of our products?
Fill out the form, our Sales Team will contact you as soon as possible.




